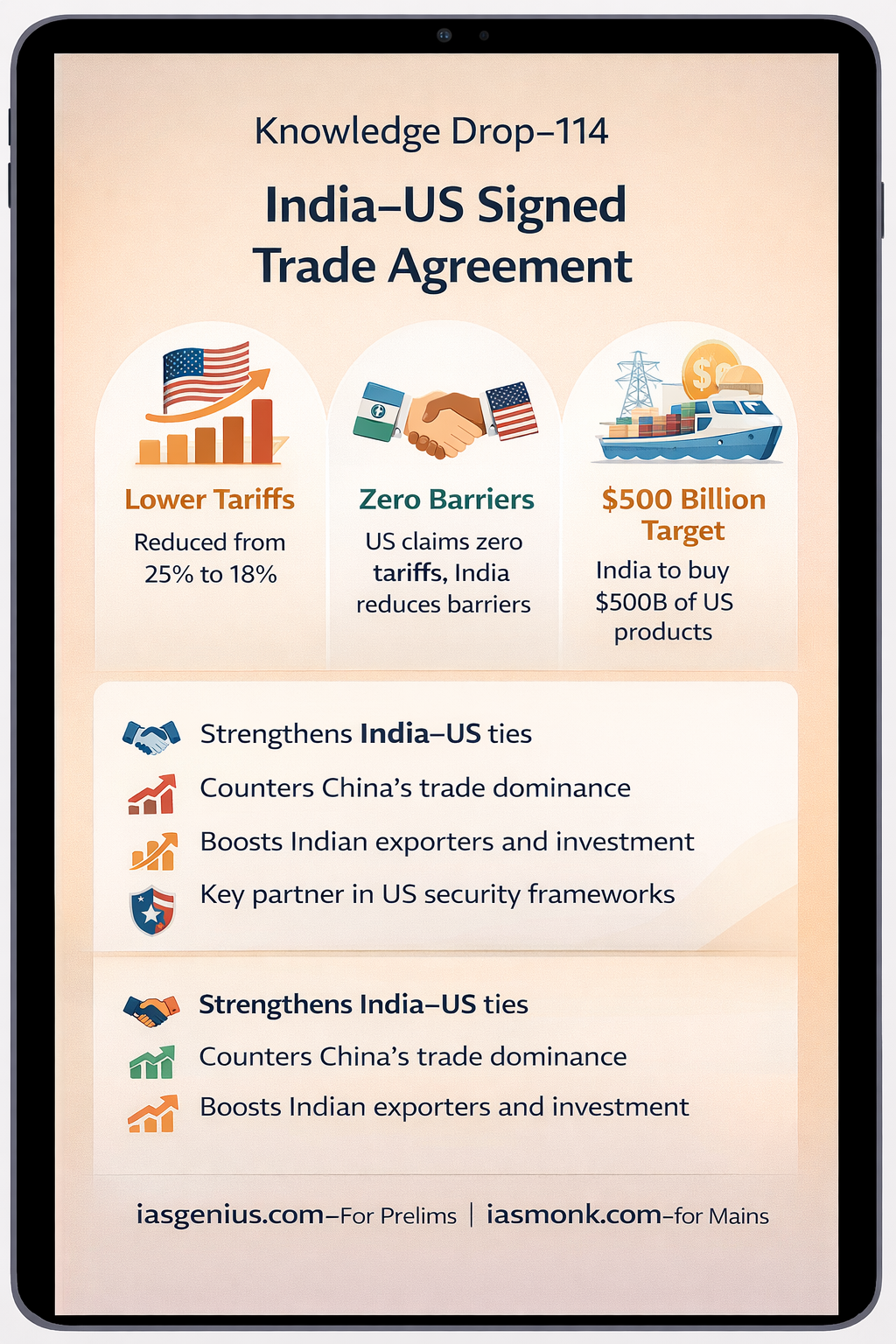
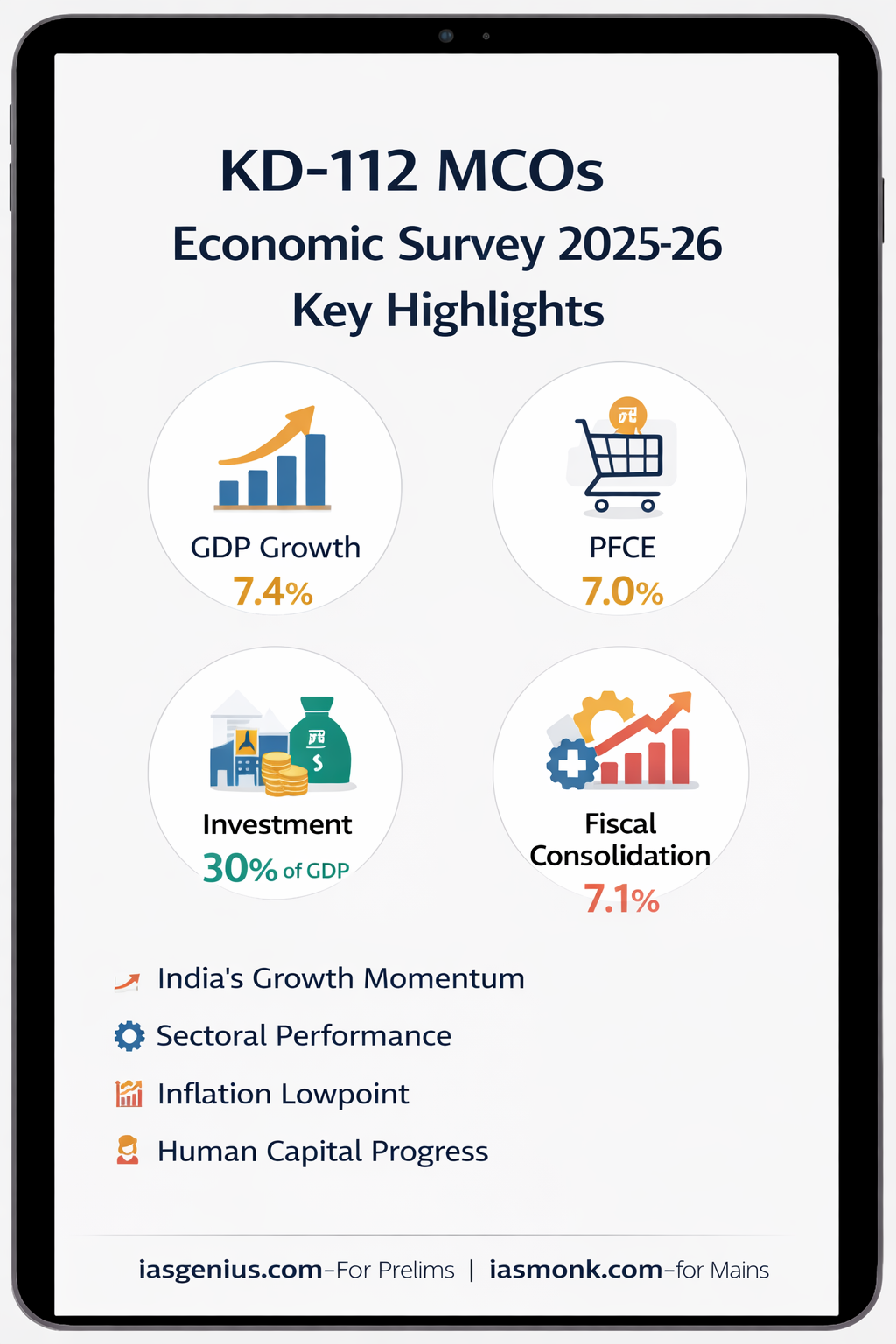
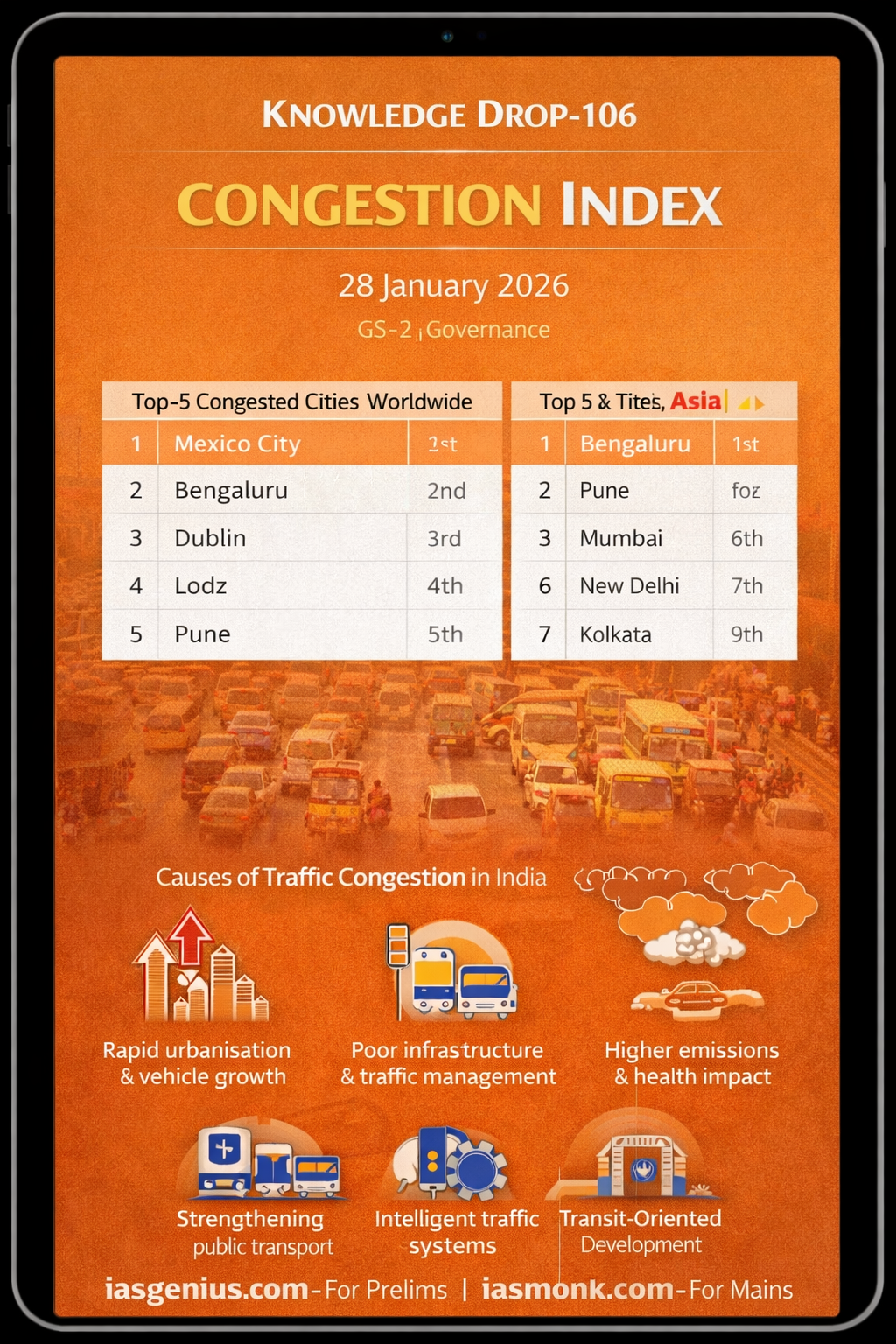
📘 Q.7 IAS Prelims 2021 — GS | Science & Technology | Why Water Is the Universal Solvent
Authentic Classroom Explanation by IAS Monk
💧 Q7. Why Water Is the Universal Solvent
📌 Question
Water can dissolve more substances than any other liquid because:
(a) it is dipolar in nature
(b) it is a good conductor of heat
(c) it has high value of specific heat
(d) it is an oxide of hydrogen
✅ Correct Answer: (a) it is dipolar in nature
🧑🏫 Authentic Classroom Explanation by IAS Monk
From first principles to final option — the UPSC way.
🔬 Concept First
Dissolution depends on electrical interactions between molecules, not on heat capacity or chemical naming.
👉 Water’s secret lies in its polarity.
🔍 Option-wise Analysis
- Option (a): Dipolar in nature ✅
- Water molecule has:
- Partial positive charge near hydrogen
- Partial negative charge near oxygen
- This polarity allows water to:
- Surround ions
- Break ionic bonds
- Dissolve polar substances
- Water molecule has:
- Option (b): Good conductor of heat ❌
- Thermal property, unrelated to solubility
- Option (c): High specific heat ❌
- Explains climate moderation, not dissolution
- Option (d): Oxide of hydrogen ❌
- Chemical classification, no explanatory power here
🔍 Curiosity Raiser
Did you know?
Water dissolves salt not by “melting” it, but by pulling ions apart using electrical forces.
🧠 Enrichment Notes (UPSC Lens)
- Like dissolves like:
- Polar solvents dissolve polar/ionic substances
- Non-polar solvents dissolve non-polar substances
- Why oil doesn’t dissolve in water:
- Oil molecules are non-polar
- Common UPSC Trap:
- Mixing thermal properties with chemical behaviour
- Exam Value:
- Foundational chemistry principle with high repeatability
🪶 IAS Monk Closing Whisper
Water dissolves the world because it carries hidden charges.